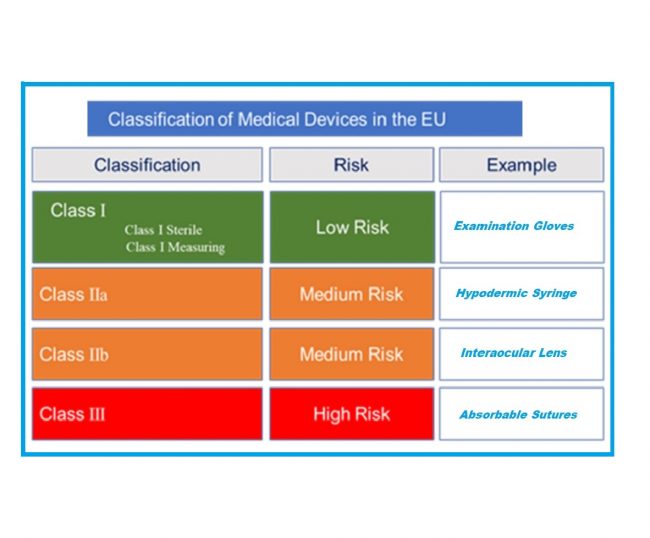
Do Toys Need a CE Mark? Detailed Overview
Learn why toys need CE mark and what role it plays in making toys safe for use in Europe. Discover why CE marking is necessary for toys and how I3CGlobal can help with this task. Ensuring Safety: Do Toys Need a CE Mark? In the EU, the safety of people is paramount particularly for products…